Air Delivery of Super-Cooled COVID-19 Vaccines
- By : PureAire Monitoring Systems
- Posted on : December 09, 2020
- News Room
There are several potential COVID-19 vaccines that may soon be available for widespread distribution. In particular, the United Kingdom has recently approved Pfizer’s vaccine, and the U.S. Food and Drug Administration is considering extending Emergency Use Authorization to the Pfizer and Moderna vaccines.
That is certainly promising news, but storage, transportation, and delivery of these potentially game-changing vaccines will be quite challenging, with the CEO of the International Air Transport Association describing the distribution of COVID-19 vaccines as “the largest and most complex logistical exercise ever” undertaken.
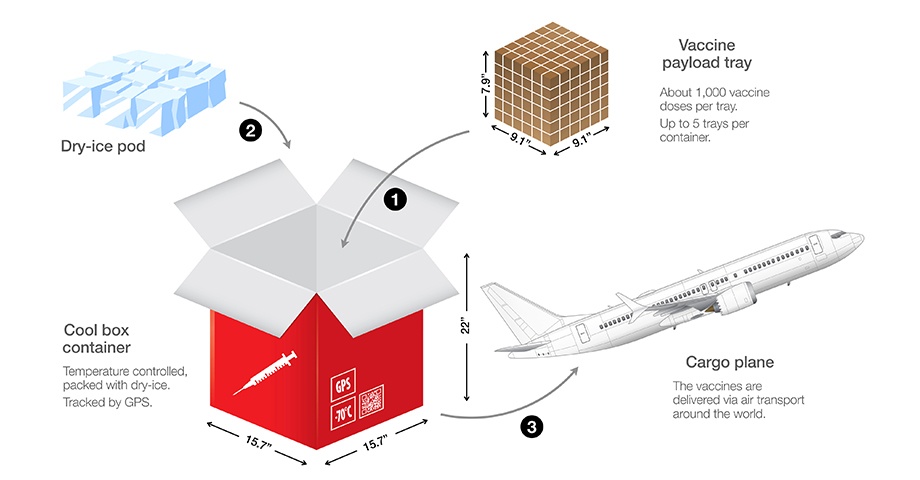
It is not just the huge numbers (literally, in the billions of doses) and vast geographic scope (worldwide, requiring delivery to every country on the planet) that make the COVID-19 vaccine distribution task so daunting, but both the Pfizer and Moderna vaccines must be stored and transported in strict climate-controlled environments (reportedly, at some -70 degrees Celsius for Pfizer, and -20 degrees Celsius for Moderna) as integral parts of the vaccines’ “cold chains.”
COVID-19 Vaccine Cold Chain
The U.S. Centers for Disease Control (the “CDC”) describes a cold chain as a temperature-controlled supply chain that includes all vaccine-related equipment and procedures. The vaccine cold chain begins with a cold storage unit at the vaccine manufacturing plant, extends to the transport and delivery of the vaccine (including proper storage at the provider facility), and ends with the administration of the vaccine to the patient. A breakdown in protocols anywhere along the cold chain could reduce the effectiveness of, or even destroy, a vaccine.
Given the extreme cold temperatures required within their cold chains by the Pfizer and Moderna vaccines (and, perhaps, other COVID-19 vaccines that may now be under development by other firms), various companies within the vaccine delivery network (including temperature-controlled container manufacturers, logistics specialists, storage facility operators, commercial airlines, and dry ice producers) have been hard at work for months to meet the challenges associated with safely storing and transporting billions of vaccine doses once, as now appears to be at hand, they finally become available for international distribution.
Creating Super-Cold Environments
Dry ice, which is the common name for solid (i.e., frozen) carbon dioxide, is often used in cold chains to maintain the very cold temperatures required to keep certain vaccines viable. At a temperature of approximately -78.5 degrees Celsius (equating to -109.3 degrees Fahrenheit), dry ice is significantly colder than frozen water (that is, conventional ice), making it ideal for transport and storage of those vaccines which require an extremely cold temperature environment.
Safety precautions are critical when shippers use dry ice in the transportation and storage of vaccines. Unlike conventional ice, dry ice does not melt into a liquid. Instead, dry ice “sublimates” (changes from a solid to a gas state), turning into carbon dioxide gas. In poorly ventilated, confined spaces, such as storage rooms, railway cars, trucks, and cargo holds in airplanes, carbon dioxide can build up, creating a potentially serious health risk to transportation workers, including ground and flight crews.
Certain vaccine manufacturers may elect to ship their vaccines in multi-layered, storage canisters chilled with liquid nitrogen, rather than dry ice. We note that the potential health risks associated with nitrogen leaks are similar to those that may be caused by dry ice sublimation.
Oxygen Deficiency Risks Associated with Super-Cooled Environments
Carbon dioxide (as is nitrogen) is an oxygen-depleting gas that is both odorless and colorless. As such, absent appropriate monitoring, personnel working with the transportation of COVID-19 and other vaccines kept frozen with dry ice or liquid nitrogen likely would be unable to detect if dry ice were to sublimate (causing CO2 levels to rise), or if there were a nitrogen gas leak, and an associated decrease in oxygen.
According to the Occupational Safety and Health Administration (OSHA), an environment in which oxygen levels fall below 19.5 percent is considered an oxygen-deficient atmosphere and should be treated as immediately dangerous to health or life. When there is not enough oxygen in the air, persons working in the affected area may become disoriented, lose consciousness, or even suffocate due to the lack of sufficient oxygen.
FAA Guidance/Increased Air Shipment Capacity/Risk Mitigation
On May 22, 2009, the U.S. Federal Aviation Administration (the “FAA”) issued Advisory Circular No. 91-76A to specifically address the risks associated with the sublimation of dry ice aboard aircraft and, historically, the FAA has permitted even widebody aircraft to carry only relatively small amounts (typically not exceeding 1-1.5 tons per flight) of dry ice in refrigerated and insulated containers.
However, The Wall Street Journal (the “WSJ”) reported on November 29, 2020, that, in order to maintain the ultra-cold temperatures required by Pfizer’s COVID-19 vaccine, United Airlines has recently sought, and obtained, FAA approval to carry up to 15, 000 pounds (7.5 tons) of dry ice per flight. In a December 2, 2020 interview with CNN, Josh Earnest, Chief Communications Officer with United Airlines, noted that the FAA approval will allow United to ship as many as 1.1 million doses of COVID-19 vaccines on each flight of its commercial 777 airplanes.
Notwithstanding the FAA’s relaxation of dry ice weight limits to permit United Airlines to help bring the COVID-19 pandemic under control, it remains focused on risks associated with air shipments of dry ice. In its November 29, 2020 reporting, the WSJ noted that “regulators restrict the amount of dry ice that can be carried on passenger jets because they typically lack the equipment to monitor and mitigate any leaked carbon dioxide.”
Fortunately, by utilizing a top-quality oxygen-deficiency monitor, vaccine storage and transportation personnel, including flight crews, can safely track levels of oxygen and detect (and react to) potentially dangerous low oxygen levels, whether caused by dry ice sublimation or a nitrogen gas leak.
PureAire Monitoring Systems, Inc.
 PureAire Monitoring Systems’ Oxygen Deficiency Monitor offers thorough air monitoring, with no time-consuming maintenance or calibration required. A screen displays current oxygen levels, for at-a-glance reading by crew members, who derive peace of mind from the Monitor’s presence and reliable performance.
PureAire Monitoring Systems’ Oxygen Deficiency Monitor offers thorough air monitoring, with no time-consuming maintenance or calibration required. A screen displays current oxygen levels, for at-a-glance reading by crew members, who derive peace of mind from the Monitor’s presence and reliable performance.
Built with zirconium oxide sensor cells, to ensure longevity, the Monitor can last, trouble-free for 10 years in normal working conditions.
Our Oxygen Deficiency Monitor does not rely on the partial pressure of oxygen to operate, meaning that the Monitor is not affected by the changing pressure inside an aircraft due to altitude changes. In the event that dry ice begins to sublimate (causing carbon dioxide levels to rise), or if there is a nitrogen leak, and oxygen decreases to unsafe levels, PureAire’s Monitor will set off an alarm, complete with horns and flashing lights, alerting flight personnel to take corrective action.
For over 20 years, PureAire Monitoring Systems has been an industry leader in manufacturing long-lasting, accurate, and reliable Oxygen Deficiency Monitors. We have dedicated ourselves to ensuring the safety and satisfaction of our clients, many of which have very sophisticated operating requirements. We are proud to note that NASA’s SOFIA-Stratospheric Observatory for Infrared Astronomy–a Boeing 747SP aircraft modified to carry a 2.7 meter (106 inch) reflecting telescope–carries onboard a PureAire Oxygen Deficiency Monitor.