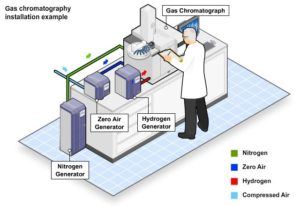
GAS CHROMATOGRAPHY
To separate chemical compounds for analysis — whether in forensic science or medical research — a process known as gas chromatography (GC) is used. This process relies on carrier gases to pass over the substance being analyzed and separate the material into its component parts for identification. To ensure the analysis is accurate, inert gases, including argon, nitrogen, and helium, are used as carrier gases. These gases do not react with substances under analysis.
Argon, helium, and nitrogen are all safe to use; however, all react with oxygen by displacing oxygen molecules. If argon gas were to leak from the supply lines, it would displace room air and cause respiratory distress and death via asphyxiation for employees. Since argon has no discernible color or odor, there would be no way for employees to tell that a gas leak had occurred.
Gas chromatography facilities can protect workers from leaks of carrier gases by using oxygen monitors to track levels of oxygen in the room. The monitors go off when oxygen falls too low, providing sufficient notice for employees to evacuate before suffering respiratory problems. For more information on the use and storage of gases used in gas chromatography locations, please see — NIH -Protocol for Use and Maintenance of Oxygen Monitoring Devices.